Associated Faculty
Petru Andrei
Hanwei Gao
Daniel Hallinan
Kenneth Hanson
Yan-Yan Hu
Susan Latturner
Biwu Ma
Subramanian (Rama) Ramakrishnan
Per Arne Rikvold
Michael Shatruk
Jim Zheng
Jose L. Mendoza-Cortes
Research Examples
Li-ion and Li-air batteries
Li-ion batteries are the most commonly used energy batteries in portable electronics and more recently are becoming more an more prominent in military, electrical vehicle, and aerospace applications.

There is a constant interest in developing battery cells with high energy density storage. This interest is driven not only by industries requiring high electric power such as the electric car industry who requires fundamental improvements in the current Li-ion technology in order to extend the driving range of current electric vehicles, but also by many low-power electronics industries which require light, high-capacity energy storage devices. Li-air batteries have the potential to become one of the best candidates for both industries because of their high theoretical energy density, which is almost one order of magnitude larger than that of Li-ion batteries.
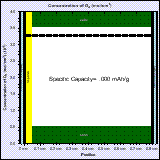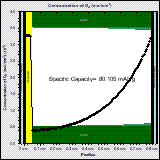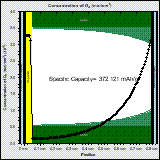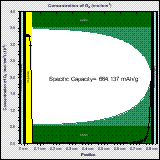
The high capacity of Li-air batteries is partly due to the fact that the active material is not stored in the battery but is taken from the atmosphere. Computer-aided-design can help designing Li-air batteries in order to improve the power density and cyclability of these batteries.
Depending on the type of the electrolyte that they use, Li-air batteries can be divided in Li-air batteries with organic electrolyte and Li-air batteries with dual electrolyte (i.e. organic electrolyte at the anode and aqueous electrolyte at the cathode). In the case of Li-air batteries with organic electrolyte the discharge product deposits on the surface of the carbon, while in the case of the batteries with dual electrolyte the discharge product is soluble in the water at the cathode. This fact makes Li-air batteries with dual electrolyte very attractive because one can use air electrodes having a fuel cell structure to continuously eliminate the discharge product from the cathode.
Our research focuses on prototyping and studding experimentally and theoretically Li-air batteries with organic and dual electrolytes.
Fuel cells
Fuel cells are used to the convert chemical energy from fuel to electricity. Our research focuses mostly on the study of proton exchange membrane fuel cells (PEMFC) which are perhaps the most common fuels cells based oxygen-hydrogen reaction.
We are currently building physics-based simulations packages (see RandFlux ) to analyze the effect of the microscopic and macroscopic structure and catalyst on the polarization curves and impendace spectra. We are developing new advanced models for the diagnosis of the state of fuel cells based on impedance spectra analysis and leakage measurements.
Supercapacitors
Supercapacitors are energy storage devices widely used in vehicle and other applications that require high power density. The power density of supercapacitors is at least one order of magnitude larger than that of conventional batteries, however, they have a slightly lower energy density.
